"Thermodynamics is a funny subject. The first time you go through it, you don't understand it at all. The second time you go through it, you think you understand it, except for one or two small points. The third time you go through it, you know you don't understand it, but by that time you are so used to it, it doesn't bother you any more."
Thermodynamics is a subset of Russell's Eighteen Attributes or Dimensions. Thermodynamics played a large role in the experimental works of John Keely.
Thermodynamics is a physical science that studies the effects on material bodies, and on radiation in regions of space, of transfer of heat and of work done on or by the bodies or radiation. It interrelates macroscopic variables, such as temperature, volume and pressure, which describe physical properties of material bodies and radiation, which in this science are called thermodynamic systems.
Historically, thermodynamics developed out of a desire to increase the efficiency of early steam engines, particularly through the work of French physicist Nicolas Leonard Sadi Carnot (1824) who believed that the efficiency of heat engines was the key that could help France win the Napoleonic Wars. Scottish physicist Lord Kelvin was the first to formulate a concise definition of thermodynamics in 1854:
"Thermodynamics is the subject of the relation of heat to forces acting between contiguous parts of bodies, and the relation of heat to electrical agency."
Initially, the thermodynamics of heat engines concerned mainly the thermal properties of their 'working materials', such as steam. This concern was then linked to the study of energy transfers in chemical processes, for example to the investigation, published in 1840, of the heats of chemical reactions by Germain Hess, which was not originally explicitly concerned with the relation between energy exchanges by heat and work. Chemical thermodynamics studies the role of entropy in chemical reactions. Also, statistical thermodynamics, or statistical mechanics, gave explanations of macroscopic thermodynamics by statistical predictions of the collective motion of particles based on the mechanics of their microscopic behavior.
Thermodynamics describes how systems change when they interact with one another or with their surroundings. This can be applied to a wide variety of topics in science and engineering, such as engines, phase transitions, chemical reactions, transport phenomena, and even black holes. The results of thermodynamics are essential for other fields of physics and for chemistry, chemical engineering, aerospace engineering, mechanical engineering, cell biology, biomedical engineering, materials science, and are useful for other fields such as economics.
Many of the empirical facts of thermodynamics are comprehended in its four laws.
- The first law specifies that energy can be exchanged between physical systems as heat and thermodynamic work.
- The second law concerns a quantity called entropy, that expresses limitations, arising from what is known as irreversibility, on the amount of thermodynamic work that can be delivered to an external system by a thermodynamic process.
Many writers offer various axiomatic formulations of thermodynamics, as if it were a completed subject, but non-equilibrium processes continue to make difficulties for it. Wikipedia, Thermodynamics
See Also
Carnot Cycle
Carnot Theorem
differential densities
Disturbance of Equilibrium
Figure 12.09 - Dimensions and Relationships
Figure 12.13 - Some Multi-Dimensional as Inverse and Direct Reciprocal Relationships
First Law of Thermodynamics
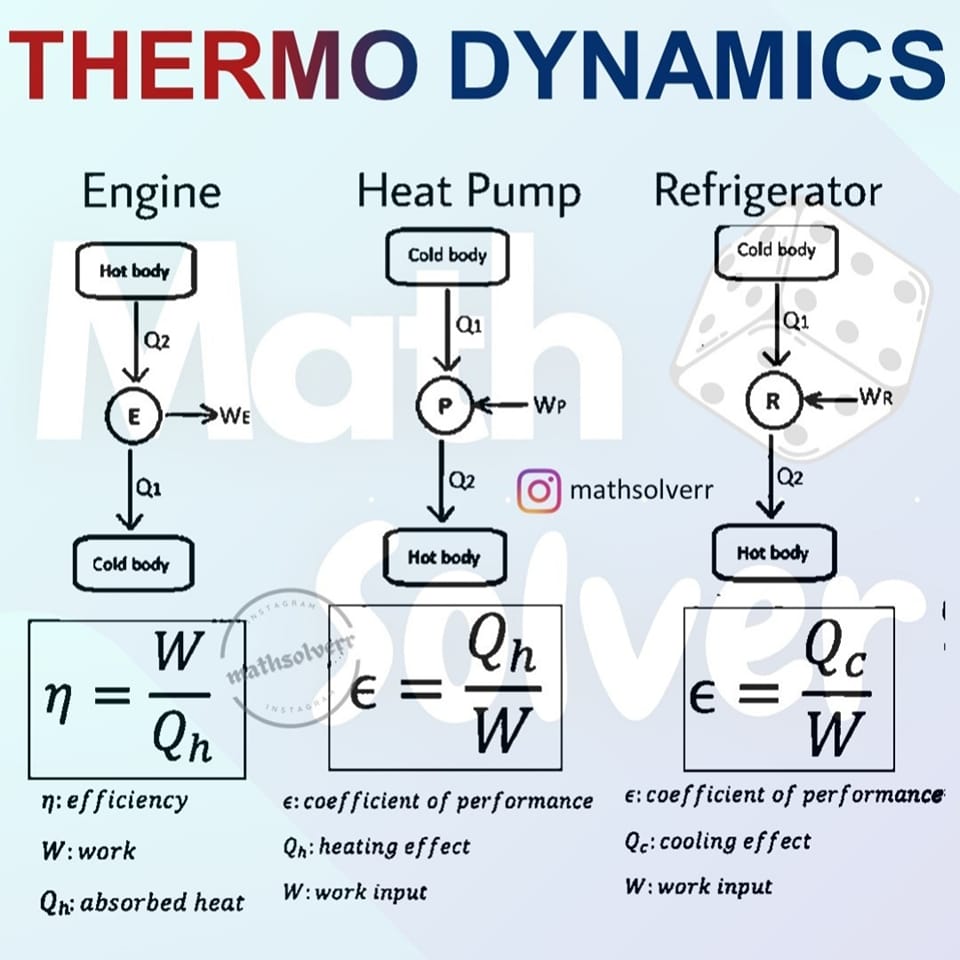
Heat Engine
LAW OF THERMODYNAMICS
Laws of Thermodynamics
Russells Laws of Thermodynamics
Russell Wavefunction
Second Law of Thermodynamics
State
thermodynamic equilibrium
Thermopedia - The Most Reliable Source for Thermodynamics, Heat Transfer, Fluid Flow Science and Technologies
Third Law of Thermodynamics
Wavefunction
12.11 - Eighteen Attributes or Dimensions
16.13 - Differential Densities
2.7 - Differential Densities
3.6 - Differential Densities Begin to Form
8.32 - Electroacoustic Thermodynamic Transduction
